A potentially serious issue with a batch of antidepressant medication has been withdrawn due to a manufacturing error.
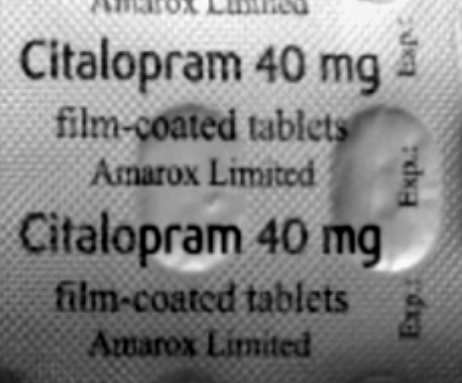
Amarox Limited is recalling one batch of Sertraline 100mg film-coated tablets as a precautionary measure due to a manufacturing error that led to two antidepressant medicines being packaged incorrectly, the recall was followed up due to a patient complaining that a pack of Sertraline 100mg tablets contained Citalopram 40mg tablets inside the sealed carton.
Affected batch number is V2500425 with expiry date 05/2028 they were first distributed on 28/11/25
Sertraline and citalopram are both selective serotonin reuptake inhibitors (SSRIs) used to treat depression, anxiety disorders, and related mental health conditions by boosting brain serotonin.
Patients who believe they have already taken any Citalopram 40mg tablets by mistake or are experiencing side effects, are advised to seek medical advice immediately, most at risk of issues are those under 18 and over 60-year-old’s
Dr Alison Cave, MHRA Chief Safety Officer, said:
“If you have been prescribed Sertraline 100mg tablets and have received batch number V2500425, please check the carton contains the right medication. You can find the batch number and expiry date printed on the side of the outer packaging.
“If the blister strips inside the carton are labelled Citalopram 40mg, please contact your pharmacy as soon as possible. If they are labelled Sertraline 100mg, no further action is needed.”
“Patients who have accidentally taken citalopram instead of – or as well as – sertraline, may experience some heightened serotonergic side effects. These can include nausea, headache, sleep changes, and mild anxiety.”
Pharmacists, or any other healthcare professionals involved in dispensing should identify and contact any patients who may have been dispensed the impacted product and request it be returned if they have any remaining medicine.
If any patients are identified with this product, pharmacists and other healthcare professionals involved in dispensing should contact the patients’ GP, or clinician responsible for the care of the patient, to discuss treatment review and whether a new prescription is required for ongoing resupply.
Patients may need to be monitored by their doctor or another healthcare professional, particularly if they are over 65 or under 18, have cardiac or liver conditions, or have been told that their body processes certain medicines differently.
Any suspected adverse reactions should also be reported via the MHRA Yellow Card scheme.
The MHRA has advised healthcare professionals to stop supplying the affected batch and return all remaining stock to their suppliers.